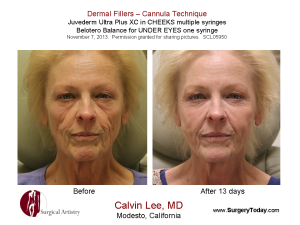
BOTOX® Medical and BOTOX® Cosmetic
The BOTOX formulations are the same. But the uses are different. As the terms say, BOTOX medical is used for Medical purposes. And BOTOX Cosmetic is used for cosmetic purposes. There are many OFF-LABEL FDA uses of BOTOX, but here we explore the ON-LABEL uses for medical problems and cosmetic problems.
Medical uses of BOTOX
Sometimes called BOTOX THERAPEUTIC
More specifically, medical uses of BOTOX which are ON-LABEL FDA:
- BOTOX for Overactive bladder symptoms
- BOTOX for Urine Incontinence due to neurological disease
- BOTOX for Headaches, specifically chronic migraines (15 or more days each month, longer than 4 hours each day)
- BOTOX for upper limb spasticity
- BOTOX for cervical dystonia
- BOTOX for strabismus: an eye muscle problem
- BOTOX for blepharospasm: an abnormal spasm of eyelids.
- BOTOX for axillary hyperhidrosis
BOTOX Injection sites for Blepharospasm
The BOTOX injection site picture from Allergan. BOTOX has been the main blepharospasm treatment since FDA approval in 1989.

Cosmetic uses for BOTOX
- Glabellar lines = Frown lines between the eyebrows. Botox was approved for usage for cosmetically reducing the appearance of wrinkles, temporarily between the eyebrows in patients between the age of 18 to 65 years old. This was approved by the FDA in 2002.
- Crows feet = Lines on the side of the eyes. In September 2013, the FDA approved Botox for use in the crows feet area. Botox for the Crows Feet – a entry in my Modesto Botox Blog
Again all the above mentions are FDA approved – ON LABEL usage. There are other medical and cosmetic uses of BOTOX but they require a physician to determine the possibility of OFF LABEL-FDA use.
Differences in Dosages in Medical Botox vs. Cosmetic Botox
Usually many more Botox units are needed for medical indications than for cosmetic indications.
Disclaimer for this BOTOX webpage:
Unless you are directly looking at my actual crows feet and my glabellar lines, there is no establishment of a doctor-patient relationship. Please see your own doctor’s wrinkles in person so that you can get medical advice. Above is for information only – not to be considered any sort of medical advice. Furthermore, looking at a picture or video of my crows feet and glabellar wrinkles does not count – it has to be a live and in-person viewing.